Who We Are
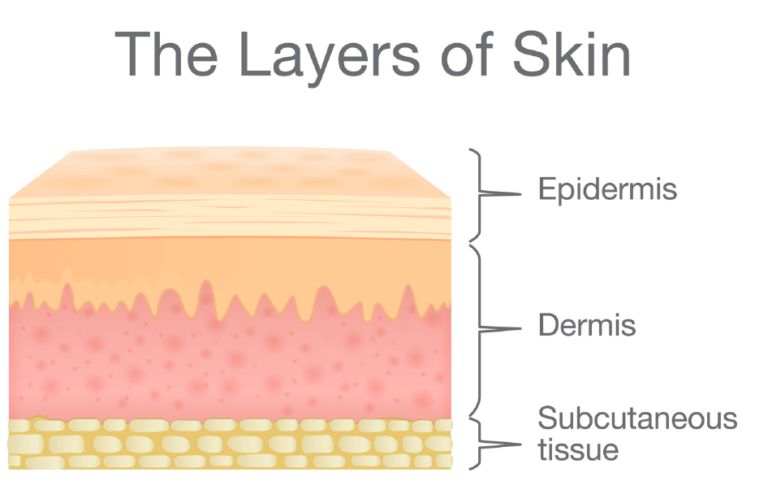
Ethoderm is a biotechnology company that is focused on delivering therapeutics transdermally. This method is minimally invasive and it is cost-effective. Our core focus is on delivering small molecules and peptides that impact the symptoms associated with chronic diseases and have minimal side effects.